Neptunium
93
Np
Grupp
Ej tillämplig
Period
7
Block
f
Protoner
Elektroner
Neutroner
93
93
144
Generella Egenskaper
Atomnummer
93
Atommassa
[237]
Masstal
237
Kategori
Aktinider
Färg
Silver
Radioaktiv
Ja
Named after the planet Neptune
Kristallstruktur
Enkelt ortorombiskt
Historia
Neptunium was the first synthetic transuranium element of the actinide series to be discovered.
Neptunium was first produced by Edwin McMillan and Philip H. Abelson in 1940 at Berkeley Radiation Laboratory of the University of California.
The team produced the neptunium isotope 239Np by bombarding uranium with slow moving neutrons.
Neptunium was first produced by Edwin McMillan and Philip H. Abelson in 1940 at Berkeley Radiation Laboratory of the University of California.
The team produced the neptunium isotope 239Np by bombarding uranium with slow moving neutrons.
Elektroner per skal
2, 8, 18, 32, 22, 9, 2
Elektronkonfiguration
[Rn] 5f4 6d1 7s2
Neptunium is obtained as a by-product from nuclear reactors
Fysikaliska Egenskaper
Aggregationstillstånd
Fast
Densitet
20,45 g/cm3
Smältpunkt
910,15 K | 637 °C | 1178,6 °F
Kokpunkt
4273,15 K | 4000 °C | 7232 °F
Smältvärme
10 kJ/mol
Ångbildningsvärme
335 kJ/mol
Specifik värmekapacitet
- J/g·K
Förekomst i jordskorpan
Ej tillämplig
Förekomst i universum
Ej tillämplig
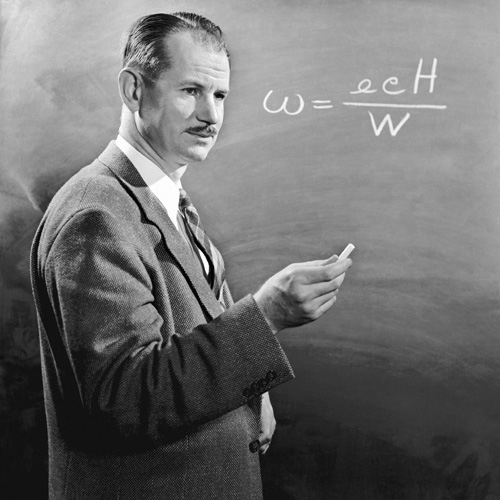
CAS-nummer
7439-99-8
PubChem CID-nummer
Ej tillämplig
Atomära Egenskaper
Atomradie
155 pm
Kovalent radie
190 pm
Elektronegativitet
1,36 (Paulingskalan)
Jonisationspotential
6,2657 eV
Molvolym
11,62 cm3/mol
Värmeledningsförmåga
0,063 W/cm·K
Oxidationstillstånd
3, 4, 5, 6, 7
Användningsområden
Neptunium is used mainly for research purposes.
When bombarded with neutrons 237Np is used to produce 238Pu which is used for spacecraft generators and terrestrial navigation beacons.
237Np is used in devices for detecting high-energy neutrons.
When bombarded with neutrons 237Np is used to produce 238Pu which is used for spacecraft generators and terrestrial navigation beacons.
237Np is used in devices for detecting high-energy neutrons.
Neptunium is harmful due to its radioactivity
Isotoper
Stabila isotoper
-Instabila isotoper
225Np, 226Np, 227Np, 228Np, 229Np, 230Np, 231Np, 232Np, 233Np, 234Np, 235Np, 236Np, 237Np, 238Np, 239Np, 240Np, 241Np, 242Np, 243Np, 244Np